Prof. Dr. rer. nat. Julia von Maltzahn is FGW Professor for Stem Cell Biology of Aging at the Brandenburg University of Technology Cottbus-Senftenberg (BTU).
The research group stem cell biology of aging investigates alterations in muscle stem cell which occur during aging and during differentiation. Furthermore, they study the causes for impaired myogenic differentiation in rhabdomyosarcoma.
The final goal is the improvement of regeneration of skeletal muscle in age and disease.
Location: B-TU Cottbus-Senftenberg, Universitätsplatz 1, 01968 Senftenberg, Office 14E.402
Skeletal muscle fulfills a multitude of functions in the organism, among them voluntary movements. Furthermore, skeletal muscle has a remarkable ability to adapt and to regenerate. For instance, the size of myofibers – the units which are responsible for the movement of skeletal muscle – can be adapted due to physical exercise or disuse. With increasing age, plasticity of skeletal muscle is reduced concomitant with an altered ability to regenerate.
Muscle stem cells, also called satellite cells, are essential for regeneration of skeletal muscle. Under resting conditions muscle stem cells are quiescent but are activated due to increased muscle activity or damage of skeletal muscle. During regeneration of skeletal muscle the cells proliferate and undergo myogenic differentiation and subsequent fusion resulting in the formation of newly formed myofibers. During aging muscle stem cells and their immediate niche undergo alterations.
Our research group investigates those intrinsic alterations and alterations in the immediate muscle stem cell niche with the aim to identify signaling pathways which are altered during aging to rejuvenate aged muscle stem cells. The final goal of our work is the restoration of homeostasis of muscle stem cells and the treatment of sarcopenia, the age related loss of muscle mass and function.
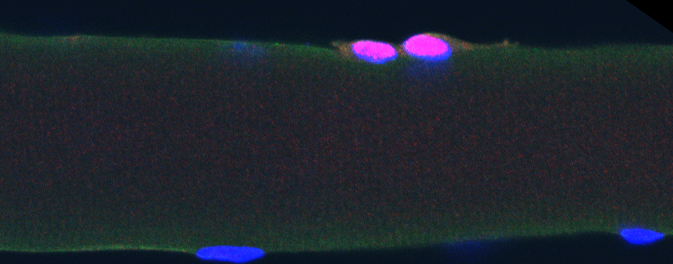
Muscle stem cells on a myofiber (Pax7 – marker for muscle stem cells in red, Dapi in blue)
We are investigating intrinsic alterations of muscle stem cells and their immediate niche which occur during aging. Thereby we aim for a better understanding of alterations which occur during aging to help develop therapies to improve regeneration of skeletal muscle in the aged.
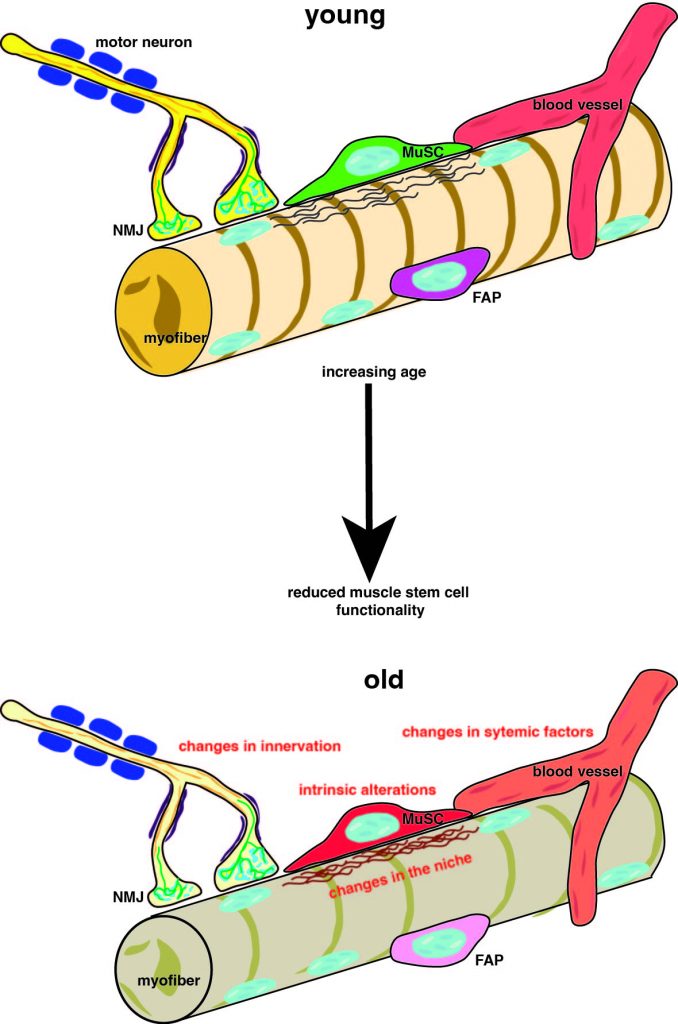
Alterations in muscle stem cells during aging. (modified after Henze et al., 2020)
It is known that induction of myogenic differentiation is impaired during aging. We aim to understand the molecular causes for this reduced ability to differentiate with the goal to improve induction of differentiation in the aged.
With increasing age also the likelihood of cancer increases. A large proportion of cancer patients is suffering from cancer cachexia, the cancer related loss of muscle mass and function. Cancer cachexia is associated with a reduced ability to regenerate and impairments in differentiation of muscle stem cells. We are investigating the signaling pathways and molecular mechanisms which cause this impaired differentiation. Thereby we aim to support the development of new therapies to improve regeneration of skeletal muscle in cancer cachexia.
Rhabdomyosarcomas are soft tissue sarcomas which mostly manifest in children. They are thought to derive from muscle progenitor cells. Rhabdomyosarcomas can be divided in two major subgroups, the alveolar and the embryonal rhabdomyosarcomas. All rhabdomyosarcoma cells share an impairment in myogenic differentiation. We are investigating the molecular causes for this impaired myogenic differentiation in alveolar and embryonal rhabdomyosarcoma. Through gaining a better understanding of the molecular causes for this impaired myogenic differentiation we want to support the development of therapies of rhabdomyosarcomas. Here, we focus on the induction of myogenic differentiation into postmitotic muscle cells.
Schmidt, M., Poser, C., Janster, C., and von Maltzahn, J. (2022). The hairpin region of WNT7A is sufficient for binding to the Frizzled7 receptor and to elicit signaling in myogenic cells. Comput Struct Biotechnol J 20, 6348-6359. 10.1016/j.csbj.2022.10.047.
Schmidt, M., Weidemann, A., Poser, C., Bigot, A., and von Maltzahn, J. (2021). Stimulation of Non-canonical NF-kappaB Through Lymphotoxin-beta-Receptor Impairs Myogenic Differentiation and Regeneration of Skeletal Muscle. Front Cell Dev Biol 9, 721543. 10.3389/fcell.2021.721543.
Schuler, S.C., Kirkpatrick, J.M., Schmidt, M., Santinha, D., Koch, P., Di Sanzo, S., Cirri, E., Hemberg, M., Ori, A., and von Maltzahn, J. (2021). Extensive remodeling of the extracellular matrix during aging contributes to age-dependent impairments of muscle stem cell functionality. Cell Rep 35, 109223. 10.1016/j.celrep.2021.109223.
Schmidt, M., Poser, C., and von Maltzahn, J. (2020). Wnt7a Counteracts Cancer Cachexia. Mol Ther Oncolytics 16, 134-146. 10.1016/j.omto.2019.12.011.
Ahrens, H.E., Huettemeister, J., Schmidt, M., Kaether, C., and von Maltzahn, J. (2018). Klotho expression is a prerequisite for proper muscle stem cell function and regeneration of skeletal muscle. Skelet Muscle 8, 20. 10.1186/s13395-018-0166-x.
Mayerl, S., Schmidt, M., Doycheva, D., Darras, V.M., Huttner, S.S., Boelen, A., Visser, T.J., Kaether, C., Heuer, H., and von Maltzahn, J. (2018). Thyroid Hormone Transporters MCT8 and OATP1C1 Control Skeletal Muscle Regeneration. Stem Cell Reports 10, 1959-1974. 10.1016/j.stemcr.2018.03.021.
Lukjanenko, L., Jung, M.J., Hegde, N., Perruisseau-Carrier, C., Migliavacca, E., Rozo, M., Karaz, S., Jacot, G., Schmidt, M., Li, L., et al. (2016). Loss of fibronectin from the aged stem cell niche affects the regenerative capacity of skeletal muscle in mice. Nat Med 22, 897-905. 10.1038/nm.4126.
Schworer, S., Becker, F., Feller, C., Baig, A.H., Kober, U., Henze, H., Kraus, J.M., Xin, B., Lechel, A., Lipka, D.B., et al. (2016). Epigenetic stress responses induce muscle stem-cell ageing by Hoxa9 developmental signals. Nature 540, 428-432. 10.1038/nature20603.
Link to Pubmed with all publications by J.v.Maltzahn https://pubmed.ncbi.nlm.nih.gov/?term=von+Maltzahn+J&sort=date